|
Pembrolizumab is also approved by the FDA for the treatment of patients with locally advanced or metastatic urothelial carcinoma (mUC) who are ineligible for cisplatin-containing chemotherapy and whose tumors express PD-L1, or in patients who are ineligible for any platinum-containing chemotherapy regardless of PD-L1 status. The ongoing KEYNOTE-676 trial (NCT03711032) is evaluating pembrolizumab plus BCG in patients with high-risk NMIBC that is persistent or recurrent after BCG induction therapy. There was 1 case each of grade 3/4 adrenal insufficiency, severe skin reaction, and type 1 diabetes mellitus. 
Grade 3/4 cases of hyponatremia and arthralgia was observed in 3 and 2 patients, respectively. Grade 3 to 5 AEs, grade 3/4 TRAEs, serious AEs, and serious TRAEs were reported in 29.4%, 12.7%, 24.5%, and 7.8% of patients, respectively. Ninety-nine patients experienced ≥1 AE, and treatment-related AEs (TRAEs) were reported in 65.7% of patients. The safety profile was found to be consistent with AEs that were reported in prior studies of the PD-1 inhibitor. Higher CRs were observed in patients who were not white (55.6%) and an ECOG performance status of 1 (51.9%) they were lowest in those who had CIS with high-grade Ta (28.0%). No patients developed muscle-invasive or metastatic disease. More than half of patients (56.9%) had a PD-L1 combined positive score 12 months of durability. Patients had CIS with T1 (11.8%), CIS with high-grade Ta (24.5%), or CIS alone (63.7%). The median number of prior BCG instillations was 12 (range, 6-45). Most patients (83.3%) were male, 67.6% were white, and 73.5% had an ECOG performance status of 0. 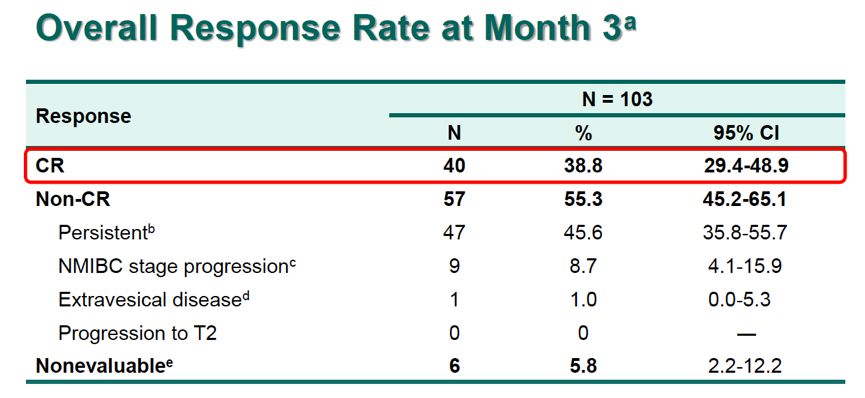
The median age was 73 years (range, 44-92), with most patients (70.6%) ≥65 years. Disease was assessed every 12 weeks, and those who did not have disease progression could receive treatment for up to 2 years. Pembrolizumab was administered at 200 mg every 3 weeks until unacceptable toxicity, persistent or recurrent high-risk NMIBC, or disease progression. In the multicenter, open-label, single-arm, multicohort, phase II KEYNOTE-057 trial, investigators enrolled 148 patients with high-risk NMIBC, 96 of whom had BCG-unresponsive, high-risk, NMIBC with CIS with or without papillary tumors. The FDA previously granted a priority review designation, also in December 2019, to the pembrolizumab NDA. 2The panel was voting that the response efficacy data compared with the toxicity findings from the KEYNOTE-057 trial demonstrate a favorable risk/benefit profile for pembrolizumab in this setting. The approval follows the December 2019 decision by the FDA’s Oncologic Drugs Advisory Committee to vote 9 to 4 supporting the approval of a new drug application (NDA) for pembrolizumab in this patient population. Additionally, 46% of responding patients experienced a CR lasting ≥12 months. 
The approval is based on findings from the phase II KEYNOTE-057 trial (NCT02625961), in which the PD-1 inhibitor elicited a complete response (CR) rate of 41% (31%-51%) in 96 patients with high-risk NMIBC with CIS with or without papillary tumors the median duration of response (DOR) was 16.2 months (0.0+, 30.4+). Pembrolizumab (Keytruda) is now FDA approved for the treatment of patients with Bacillus Calmette-Guerin (BCG)unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
